eISSN: 2578-1782 Journal Abbreviation: Therm. Sci. Eng. | Thermal Science and Engineering (TSE) is an international open access journal that publishes high-quality articles that span activities ranging from fundamental thermodynamic scientific research to the applied discussion of maximising thermodynamic efficiencies and minimising all heat losses. Topics cover thermal biology, nanotechnology, thermal energy transport, thermodynamics, thermal medical systems, and devices, etc. Interests include, are not limited to, those related to all areas of thermal science and engineering:
|
Online Submissions
Registration and login are required to submit items online and to check the status of current submissions.
Already have a Username/Password for Thermal Science and Engineering?
GO TO LOGIN
Need a Username/Password?
GO TO REGISTRATION
Submission Preparation Checklist
As part of the submission process, authors are required to check off their submission's compliance with all of the following items, and submissions may be returned to authors that do not adhere to these guidelines.
- The submission has not been previously published, nor is it under the consideration of another journal (or an explanation has been provided in Comments to the Editor).
- The submission file is in Microsoft Word format.
- Where available, URLs for the references have been provided.
- The text adheres to the stylistic and bibliographic requirements outlined in the Author Guidelines, which is found in About the Journal.
- If submitting to a peer-reviewed section of the journal, the instructions in Ensuring a Blind Review have been followed.
Privacy Statement
EnPress Publisher respects and strives to protect the privacy of its users and visitors. Hence, users and visitors are encouraged to read EnPress Publisher’s privacy policy regarding the usage and handling of user information.
(1) User information
Names and email addresses entered in all EnPress Publisher’s journal sites will be used exclusively for the stated purposes of the journals and will not be made available for any other purpose or to any other party. For submission and peer review, users should register an account for further procedures, including but not limited to name, email, address, interests, affiliation, and postcode, as editors need the information to complete in-house processes (e.g., processing a manuscript).
When users visit the publisher's website, information about the visit is saved in web logs (e.g., device, IP address, time of visit, etc.), which are only used to help improve the structure and content of the website.
(2) User rights
Users have the right to register or update their personal information and contact the publisher to cancel/delete their account if required.
(3) Third-party link
EnPress Publisher is not responsible for private information obtained by third-party websites when users log in via a pop-up screen from third-party software installed on their computer.
When users visit third-party platforms (e.g., LinkedIn, Twitter, COPE, etc.) through hyperlinks from EnPress Publisher’s journal websites, the privacy policy follows the policies of the third-party platforms.
(4) Queries or contact
For any queries about EnPress Publisher’s privacy policy, please contact the editorial office at editorial@enpress-publisher.com.
Article Processing Charges (APCs)
Thermal Science and Engineering is an Open Access Journal under EnPress Publisher. All articles published in Thermal Science and Engineering are accessible electronically from the journal website without commencing any kind of payment. In order to ensure contents are freely available and maintain publishing quality, Article Process Charges (APCs) are applicable to all authors who wish to submit their articles to the journal to cover the cost incurred in processing the manuscripts. Such cost will cover the peer-review, copyediting, typesetting, publishing, content depositing and archiving processes. Those charges are applicable only to authors who have their manuscript successfully accepted after peer-review.
| Journal Title | APCs |
|---|---|
| Thermal Science and Engineering | $500 |
We encourage authors to publish their papers with us and don’t wish the cost of article processing fees to be a barrier especially to authors from the low and lower middle income countries/regions. A range of discounts or waivers are offered to authors who are unable to pay our article processing charges. Authors can write in to apply for a waiver and requests will be considered on a case-by-case basis.
*Article No. is mandatory for payment and it can be found on the acceptance letter issued by the Editorial Office. Payment without indicating Article No. will result in processing problem and delay in article processing. Please note that payments will be processed in USD. You can make payment through Masters, Visa or UnionPay card.
Vol 8, No 2 (2025)
Table of Contents
This research investigates the effects of drying on some selected vegetables, which are Telfaria occidentalis, Amaranthu scruentus, Talinum triangulare, and Crussocephalum biafrae. These vegetables were collected fresh, sliced into smaller sizes of 0.5 cm, and dried in a convective dryer at varying temperatures of 60.0 ℃, 70.0 ℃ and 80.0 ℃ respectively, for a regulated fan speed of 1.50 ms−1, 3.00 ms−1 and 6.00 ms−1, and for a drying period of 6 h. It was discovered that the drying rate for fresh samples was 4.560 gmin−1 for Talinum triangulare, 4.390 gmin−1 for Amaranthu scruentus, 4.580 gmin−1 for Talinum triangulare, and 4.640 gmin−1 for Crussocephalum biafrae at different controlled fan speeds and regulated temperatures when the mass of the vegetable samples at each drying time was compared to the mass of the final samples dried for 6 h. The samples are considered completely dried when the drying time reaches a certain point, as indicated by the drying rate and moisture contents tending to zero. According to drying kinetics, the rate of moisture loss was extremely high during the first two hours of drying and then steadily decreased during the remaining drying duration. The rate at which moisture was removed from the vegetable samples after the drying process at varying regulated temperatures was noted to be in this trend: 80.0 ℃ > 70.0 ℃ > 60.0 ℃ and 6.0 ms−1 > 3.0 ms−1 > 1.5 ms−1 for regulated fan speed. It can be stated here that the moisture contents have significant effects on the drying rate of the samples of vegetables investigated because the drying rate decreases as the regulated temperatures increase and the moisture contents decrease. The present investigation is useful in the agricultural engineering and food engineering industries.
This study comprehensively evaluates the system performance by considering the thermodynamic and exergy analysis of hydrogen production by the water electrolysis method. Energy inputs, hydrogen and oxygen production capacities, exergy balance, and losses of the electrolyzer system were examined in detail. In the study, most of the energy losses are due to heat losses and electrochemical conversion processes. It has also been observed that increased electrical input increases the production of hydrogen and oxygen, but after a certain point, the rate of efficiency increase slows down. According to the exergy analysis, it was determined that the largest energy input of the system was electricity, hydrogen stood out as the main product, and oxygen and exergy losses were important factors affecting the system performance. The results, in line with other studies in the literature, show that the integration of advanced materials, low-resistance electrodes, heat recovery systems, and renewable energy is critical to increasing the efficiency of electrolyzer systems and minimizing energy losses. The modeling results reveal that machine learning programs have significant potential to achieve high accuracy in electrolysis performance estimation and process view. This study aims to contribute to the production of growth generation technologies and will shed light on global and technological regional decision-making for sustainable energy policies as it expands.
This study aims to investigate the enhancement in electrical efficiency of a polycrystalline photovoltaic (PV) module. The performance of a PV module primarily depends upon environmental factors like temperature, irradiance, etc. Mainly, the PV module performance depends upon the panel temperature. The performance of the PV module has an inverse relationship with temperature. The open circuit voltage of a module decreases with the increase in temperature, which consequently leads to the reduction in maximum power, efficiency, and fill factor. This study investigates the increase in the efficiency of the PV module by lowering the panel temperature with the help of water channel cooling and water-channel accompanied with forced convection. The two arrangements, namely, multi-inlet outlet and serpentine, are used to decrease the temperature of the polycrystalline PV module. Copper tubes in the form of the above arrangements are employed at the back surface of the panel. The results demonstrate that the combined technique is more efficient than the simple water-channel cooling technique owing to multi-heat dissipation and effective heat transfer, and it is concluded that the multi-inlet outlet cooling technique is more efficient than the serpentine cooling technique, which is attributed to uniform cooling over the surface and lesser pressure losses.
Metal iodide materials as novel components of thermal biological and medical systems at the interface between heat transfer techniques and therapeutic systems. Due to their outstanding heat transfer coefficients, biocompatibility, and thermally activated sensitivity, metal iodides like silver iodide (AgI), copper iodide (CuI), and cesium iodide (CsI) are considered to be useful in improving the performance of medical instruments, thermal treatment processes, and diagnostics. They are examined for their prospective applications in controlling thermal activity, local heating therapy, and smart temperature-sensitive drug carrier systems. In particular, their application in hyperthermia therapy for cancer treatment, infrared thermal imaging for diagnosis, and nano-based drug carriers points to a place for them in precision medicine. But issues of stability of materials used, biocompatibility, and control of heat—an essential factor that would give the tools the maximum clinical value—remain a challenge. The present mini-review outlines the emerging area of metal iodides and their applications in medical technologies, with a special focus on the pivotal role of these materials in enhancing non-invasive, efficient, and personalized medicine. Over time, metal iodide-based systems scouted a new era of thermal therapies and diagnostic instrumentation along with biomedical science as a whole.
Announcements
Congratulations to Prof. Qingsong Wang on Receiving the 2024 Highly Cited Award from Clarivate Analytics |
|
 |
|
| Posted: 2024-12-18 | More... |
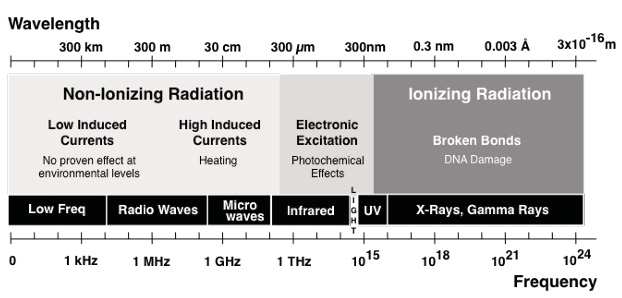
Research News: A review of Thermal effects of electromagnetic origin from heating processes to biological disturbances due to field exposure |
|
 |
|
| Posted: 2024-08-05 | More... |
New Editorial Board! |
|
The TSE's editorial team has recently undergone a reorganization and is pleased to introduce a new editorial board team. This team includes one editor-in-chief, two associate editors, and 48 editorial board members. The new editorial board will guide the editorial team in advancing the journal's success in the future. |
|
| Posted: 2024-07-01 | |
| More Announcements... |




 Open Access
Open Access
